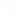How do MOFs help accelerate hydrogen production?
Researchers at IMDEA Energy have developed a new material capable of improving the production of green hydrogen by splitting water using sunlight. The work, published in the journal Journal of the American Chemical Society, opens new pathways toward more efficient and sustainable technologies for the generation of clean fuels.
Green hydrogen is considered one of the key energy vectors for the energy transition. However, its current production mainly relies on water electrolysis, a process that requires large amounts of electricity. For this reason, scientists are searching for more direct alternatives that make it possible to harness solar energy to produce hydrogen in a simpler and potentially more cost-effective way.
In this study, the research team designed a new photocatalyst based on MOFs (metal–organic frameworks), porous materials made up of metal nodes and organic molecules that can be designed with great precision at the molecular level. In particular, the researchers worked with a material previously developed at the institute, known as IEF-11, to which they applied structural engineering strategies to improve its properties.
The advance is based on two key modifications of the material. On the one hand, the researchers introduced controlled defects during microwave synthesis, which significantly increased its surface area and created new pores that facilitate the access of water molecules to the active centers of the catalyst. On the other hand, they incorporated small amounts of vanadium into the structure of the material, a process known as “doping,” which modifies its electronic properties.
Thanks to this combination of strategies, the new material is able to better absorb visible light and promote the separation of electrical charges generated during irradiation, two key factors for improving the efficiency of photocatalytic reactions.
The results show that a small amount of vanadium optimizes the catalyst’s performance, significantly increasing hydrogen production compared to the original material. In addition, the system is capable of carrying out the water-splitting reaction without the need for cocatalysts based on noble metals, which represents an important advantage for the development of more sustainable and scalable technologies.
The study also combines advanced characterization experiments with theoretical simulations to understand in detail how the structural and electronic modifications of the material influence its photocatalytic behavior.
This work demonstrates that defect engineering and metal doping can be used together to optimize the properties of MOFs and improve their performance in energy applications. The results contribute to the development of new materials capable of harnessing solar energy to produce clean fuels, an important step toward more sustainable energy systems.