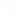Technological Offer
Photocatalytic water splitting using IMDEA-Energy Framework 11 and 13
Description
Water splitting using sun irradiation appears as a challenging process to generate solar fuels, particularly green hydrogen.
However, efficient photocatalysts are required to circumvent the energetic demand of the involved reactions. In this context, IMDEA- Energy Frameworks (IEFs) have emerged as potential platforms for hydrogen generation because the easy tunability of their electrochemical properties compared to other semiconductors. Here, we propose two new competitive porous polymers, i.e. IEF-11 and IEF- 13, showing improved performance in green hydrogen generation compared to conventional TiO2.
Advantages and innovation
This new class of porous materials, known as Metal-Organic Frameworks (MOFs) present several advantages compared to classical adsorbent and catalytic materials (carbons, zeolites, silicas):
- MOFs present versatile composition, large structural variability, very important porosity with large panel of pore sizes and shapes, easy modulation of their physicochemical properties, and are highly stable in water.
- The absorption of the solar light in the visible range (band gap) can be finely tuned by carefully selecting their synthetic conditions and composition to optimize their performance in green hydrogen generation, making them excellent candidates for water splitting compared to conventional TiO2-based semiconductors.
- IEF-11 takes advantage of the photo- and redox-active Ti4+ cation and the electroactive squarate ligand to produce the highest H2 amount reported to date for other similar polymers or semiconductors, and without the need for adding additional and expensive noble metals. This material shows an exceptional thermal and chemical stability (up to 300 ºC, pH = 10.5, under harsh conditions and aggressive chemicals) with excellent recyclability.
IEF-13 takes advantage of the redox-active Ni2+ cation and a homemade photo-/electro-active phosphonate ligand to produce H2 from water without the need for adding additional and expensive noble metals. In addition, this material shows an outstanding structural stability and robustness (up to 450 ºC, pH = 12, under harsh conditions and aggressive chemicals) with proven recyclability.
Market applications
- Water treatment plants (both, safe and wastewater): Biological Oxygen Demand (BOD).
- Research on photocatalytic generation of green hydrogen.
- Industrial reactors for hydrogen generation under sunlight or simulated sunlight during the night.
- Solar fuel production for internal combustion engines or fuel cells
Contact
Félix Marín, felix.marin@imdea.org